RefLab offers the only accredited laboratory test that supports identification of the type IIb autoimmune endotype of chronic spontaneous urticaria (CSU): the serum-induced basophil histamine release assay (s-BHRA / HR-Urticaria Test). RefLab is accredited by DANAK according to DS/EN ISO/IEC 17025:2017 (Reg. No. 472).
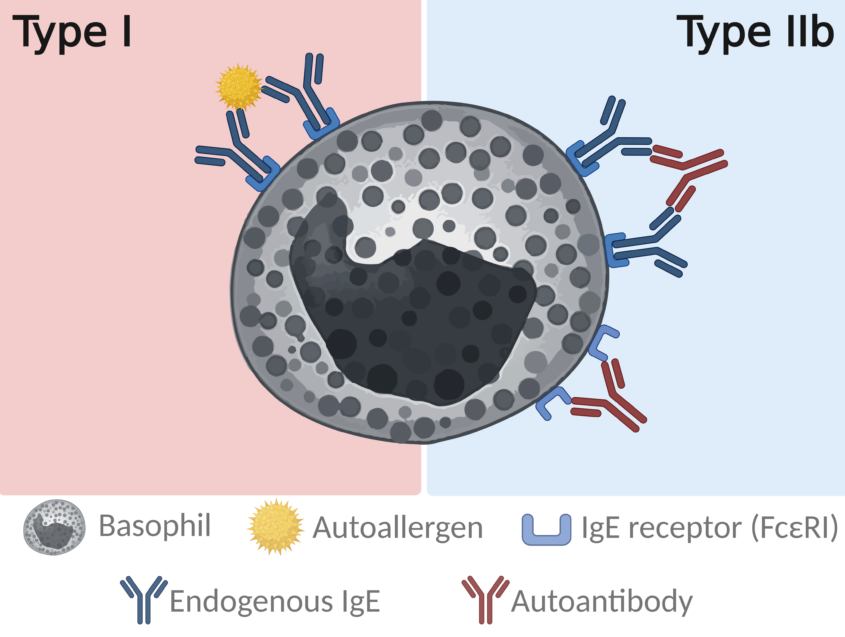
Type IIb autoimmune CSU is characterized by activating autoantibodies directed against the high-affinity IgE receptor (FcεRI) and/or IgE. Basophil-based functional tests – such as the basophil activation test (BAT) and BHRA – are used because basophils, like mast cells, express FcεRI and respond to receptor cross-linking by releasing mediators, including histamine. This makes basophil testing a practical in vitro surrogate of mast-cell activation pathways relevant to CSU. The differences between CSU patients makes it important to diagnose the patients correctly to provide them with the optimal medical care (1).
The serum-induced basophil histamine release assay (s-BHRA)
In the s-BHRA, donor basophils are challenged with patient serum to detect the presence of activating factors/autoantibodies characteristic of type IIb CSU, and the response is quantified as histamine release as depicted below.
In s-BHRA patient serum and donor basophils are incubated for 60 min at 37 °C. Histamine release (HR) is measured from stimulated and unstimulated cells, and the results are expressed as histamine release in percentage of the total histamine content.
A HR > 16.5% means that the test is positive for CSU type IIb for children as well as adult patients. A positive result identifies patients with type IIb autoimmune CSU, a distinct CSU subtype.
The test can help:
- Explain symptoms: patients learn that their hives are autoimmune and not allergy-driven, which can be reassuring.
- Guide treatment expectations: type IIb (s-BHRA–positive) patients often need higher doses of antihistamines for symptom control.
- Support screening for other autoimmune diseases where relevant, since autoantibodies are involved.
The test can be requested for children and adults by submitting a serum sample (typically 1–2 mL) to RefLab (See below). Results are reported within 7–21 business days after sample receipt.
1. Sample preparation
- The sample should be 1-2 ml plain serum in a serum seperator tube (SST).
- The serum sample can be stored up to 2 weeks at ambient room temperature.
2. Sample submission
- Complete the requisition form (print or download)
- Send the completed form via secure email to RefLab (preferred) or include a printed copy with the sample. Please use this button for sending secure email with attachments to us: Send secure email.
3. Shipping
- The sample and requisition form have to be sent in an envelope or transport container for biological samples
- Inside Europe the sample can be shipped on ambient temperature (no cooling needed)
- Outside Europe it is recommended to ship the samples frozen and preferably collecting sera from multiple patients before shipping
- Send the blood sample, requisition form (if not emailed), and any patient-supplied materials** to:
RefLab ApS
Kanalholmen 1, 2. th.
2650 Hvidovre
Denmark
