Unexpected allergic reactions are one of the reasons why drug candidates fail in the later development phase. Severe hypersensitivity or anaphylaxis can stop a program abruptly – often after significant time, cost, and animal use. RefLabs’ Pre-Clinical Drug Anaphylaxis Screening Package is designed to address this risk early, when decisions are still flexible and affordable.
Designed to de-risk development at an early stage, this package (along with all of our other services) brings together two complementary assays that evaluate the core cellular mechanisms driving anaphylaxis. Rather than relying on a single assay or preclinical animal data alone, it evaluates both major effector cell populations involved in acute allergic responses:
- Mast cells in human skin (skin microdialysis)
- Basophils in human blood (basophil histamine release assay)
By capturing both pathways, the package offers a more predictive, decision-enabling assessment of anaphylaxis liability early in development and support proof-of concept stages/experiments.
Why it works well for de-risking
Anaphylaxis is driven by rapid histamine release from immune cells. These reactions can be difficult to predict in standard toxicology studies and may not appear until late development or first-in-human dosing. This package directly measures histamine release and immune activation, making it highly relevant for early decision-making.
The results help you:
- Identify potential hypersensitivity risks early
- Compare formulations or compounds side-by-side
- Decide whether additional mitigation or reformulation is needed
- Avoid advancing high-risk candidates into costly animal or regulatory studies
Designed for clarity and confidence
The studies are performed with a small number of donors, keeping timelines and costs efficient while still generating meaningful data. This screening package is not meant to replace later safety studies—it is meant to protect them. By flagging anaphylaxis risk early, we help you move forward with greater confidence, reduce late-stage surprises, and invest your development resources where they matter most.
The preclinical drug anaphylaxis screening package – contact info@reflab.dk for more info
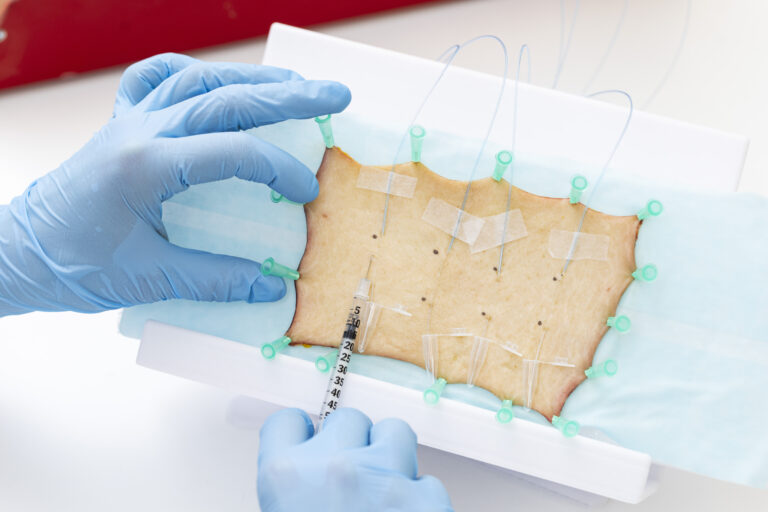
1. Skin microdialysis
A single donor is used to assess mast cell activation through microdialysis, providing direct insight into local histamine release and tissue-level hypersensitivity risk.
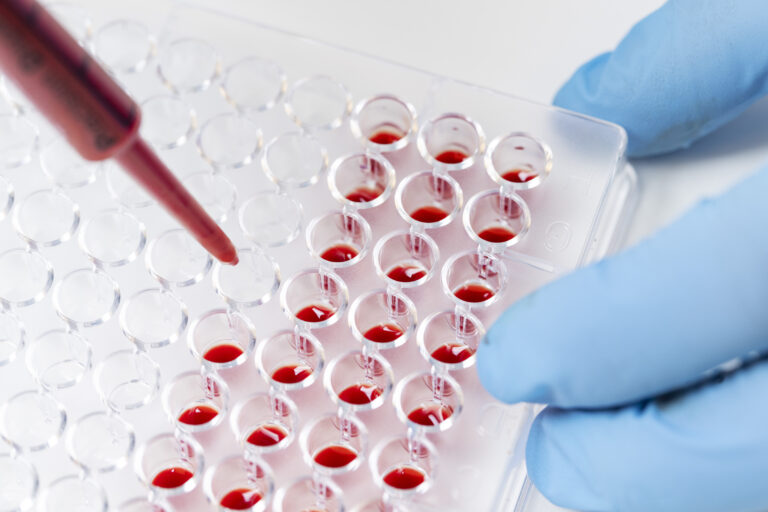
2. Basophil histamine release assay
Two independent blood donors are used to evaluate systemic basophil responses, capturing variability and strengthening confidence in early anaphylaxis risk assessment.
