RefLab supports preclinical development with specialized in vitro and ex vivo models designed to predict first-in-human immune and adverse immune responses, including pseudoallergic reactions. Our assays are tailored to new biologics, small molecules and excipients, helping you de-risk clinical entry and optimize study design.
We combine our human ex vivo skin model, a-BHRA and pharmacokinetic expertise to provide an integrated view of histamine-mediated and non-IgE–mediated mechanisms before first dosing in humans.
Ex vivo skin
Our human ex vivo skin model is a proprietary preclinical platform that uses voluntairly surgically obtained human skin tissue to evaluate histamine release and related immune activation in a clinically relevant setting. By working directly in human tissue, the model captures key aspects of skin architecture, resident immune cells and local microenvironment that are not reflected in standard cell-based assays or animal models.
The assay uses histamine release as a functional readout, allowing sensitive detection of both immune-mediated and pseudoallergic responses across a wide range of drug classes, formulations and excipients. However, other readouts are available, please contact us at info@reflab.dk for more information.
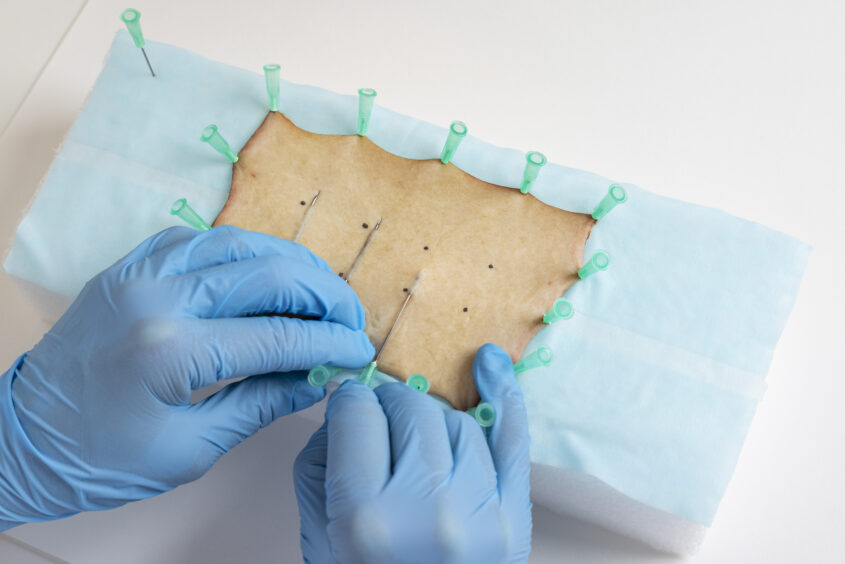
Allergen induced basophil histamine release assay
Our a-BHRA platform can be used in contract research to identify and de-risk histamine-releasing potential of new drug candidates and formulations. The assay measures functional histamine release from basophils after exposure to a test compound, providing a direct readout of immediate effector-cell activation that can complement standard in vitro safety panels.
Studies are typically run as dose–response experiments with appropriate controls and can be tailored to your program (e.g., biologics, small molecules, excipients, aggregates, or different formulations). Where relevant, the setup can be expanded to explore mechanism (e.g., IgE-dependent vs. non-IgE pathways) and to compare batches or process changes. We work with you to define clinically meaningful concentration ranges and decision criteria for candidate selection and next-step risk mitigation.
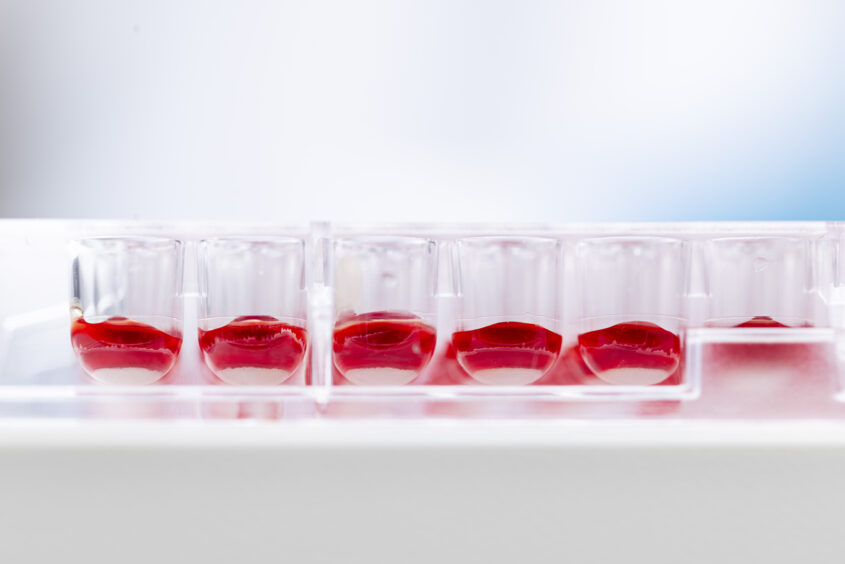
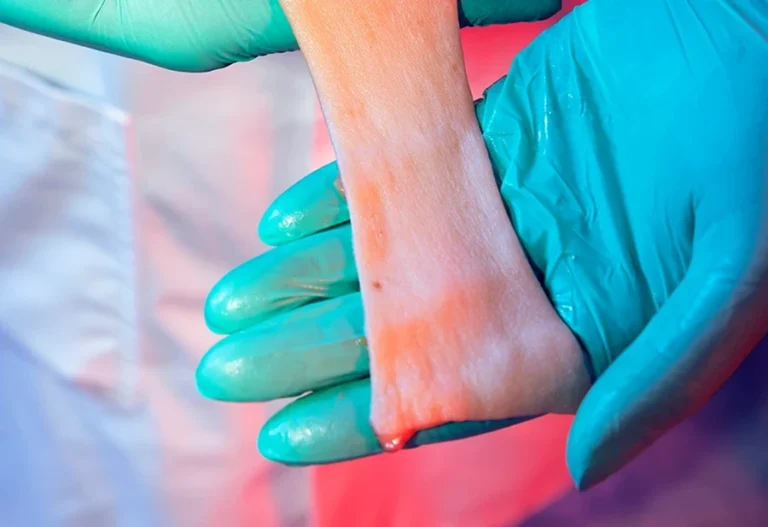
Clinical testing
RefLab offers contract analysis of human clinical samples to evaluate immune and hypersensitivity responses in the context of clinical studies. We work with commercial and academic partners who collect the samples; RefLab does not recruit or treat study participants but acts as a specialized central lab for histamine release testing.
